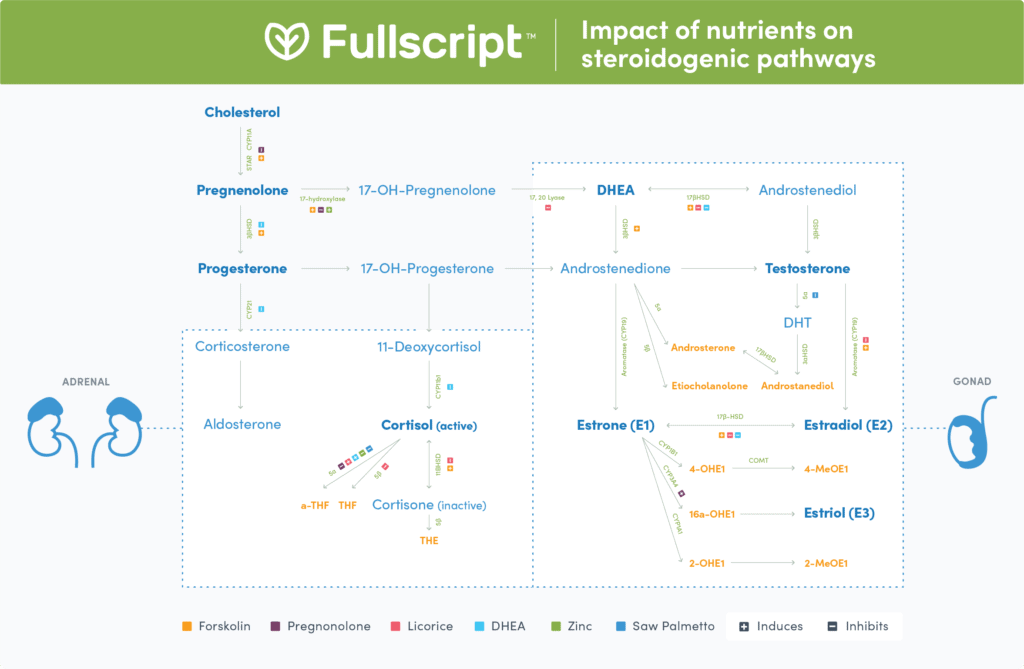
The fates of pregnenolone
While pregnenolone may exert direct hormonal effects in the body, it is also involved in three separate steroidogenic pathways, each of which produces specific hormones and metabolites:- Pregnenolone can be converted to progesterone to ultimately produce corticosterone and aldosterone
- Pregnenolone can be converted to 17-OH-progesterone and 11-Deoxycortisol to ultimately produce cortisol
- Pregnenolone can be converted to DHEA, and subsequently to testosterone, estrone, estradiol, and estriol
Pregnenolone to progesterone, corticosterone, and aldosterone
Progesterone plays a major role in pregnancy and regulation of the menstrual cycle. (27) The synthesis of progesterone from pregnenolone requires the enzyme 3βHSD, which is found primarily in the liver, brain, adrenal glands, breasts, gonads, and placenta. (53) CYP21 is responsible for the subsequent conversion of progesterone to corticosterone, which can then be converted to aldosterone via CYP11B2. (54) The primary function of aldosterone is to regulate salt and water homeostasis, which in turn regulates blood pressure. (8) CYP21 proteins are mainly found in the adrenal glands (53), though distribution has also been measured in the renal tubules, in the excretory ducts of the pancreas and salivary glands, and in the secretory ducts of the mammary glands. (66)
Start using supplements in clinical practice
Pregnenolone to cortisol
Pregnenolone and progesterone can be converted to 17-OH-pregnenolone or 17-OH-progesterone, respectively. This occurs through the use of the 17-hydroxylase enzyme found in the gonads and adrenal glands. (53) The enzyme 3βHSD can also act to convert 17-OH-pregnenolone to 17-OH-progesterone before 17-OH-progesterone undergoes further conversion to 11-deoxycortisol (54) by CYP21. (53) 11-deoxycortisol can then be converted by CYP11B1 to cortisol (53), a key hormone that acts to cause physiological and psychological changes to the body in response to stressors. (1) 11βHSD helps to regulate cortisol levels in the body by facilitating the interconversion conversion of cortisol to cortisone. By deactivating cortisol to cortisone, 11βHSD protects mineralocorticoid receptors against overstimulation by cortisol, thereby conserving receptor binding potential for mineralocorticoids such as aldosterone. (53) 11βHSD is found in the liver, testes, lungs, fat, nephrons, placenta, and colon. (53)Pregnenolone to testosterone, estrone, estradiol, and estriol
DHEA is a hormone that has been linked to cardiovascular aging, fertility, metabolism, and central nervous system (CNS) function. (61) The synthesis of DHEA from pregnenolone or progesterone requires 17-hydroxylase and 17,20 Lyase (54), which are found in the gonads and adrenal glands. (53) The 17-hydroxylase and 17,20 Lyase enzymes are active in the zona reticularis of the adrenal cortex, where DHEA and other subsequential sex hormones can be derived from pregnenolone. 17-hydroxylase is also active in the zona fasciculata. (53)
The fates of DHEA
Similarly to pregnenolone, if DHEA is not exerting direct effects on the body, it may undertake two pathways to produce steroidogenic hormones and metabolites including:- Testosterone which is subsequently converted to DHT
- Estrone, estradiol, and estriol
DHEA to testosterone and DHT
DHEA can be interconverted with androstenediol and androstenedione by 17βHSD, an enzyme which also functions to interconvert androsterone with androstanediol. Testosterone can then be produced from androstenedione via 17βHSD. 17βHSD is found in the placenta, developing follicles of granulosa cells, endometrium, testes, peroxisomes, adrenals, and prostate. (53) DHEA can also be converted to androstenedione by 3βHSD. Additionally, the 3βHSD enzyme can produce testosterone from androstenediol. (53) Testosterone has a wide variety of purposes including sexual development and function, reproductive health, and in the maintenance of muscle mass, fat metabolism, and bone density. (50) Testosterone itself may also be converted to other steroidogenic hormones. One of the primary pathways is the production of DHT facilitated by the 5α-reductase enzyme found in the skin, prostate, and liver. (78) This enzyme functions to produce androsterone by reducing androstenedione (39), but also has other substrates including progesterone, 17-OH-progesterone, cortisol, cortisone, and corticosterone. (53) DHT is the major prostatic hormone involved in prostate growth, facial hair growth, male pattern baldness, acne, and male sexual differentiation during embryogenesis. (48)DHEA to estrone, estradiol, and estriol
After DHEA is converted to androstenedione, estrogen hormones can be subsequently produced. CYP19, also known as aromatase, is the primary enzyme that converts androstenedione to estrone. CYP19 also has the ability to convert testosterone to estradiol. Aromatase is found in ovarian granulosa cells, as well as brain, fat, bone, and placenta tissues. (53) Both estrone and estradiol can be interconverted by 17βHSD. The synthesis of estrone metabolites includes the production of 2-OHE1 by CYP1A1 or the production of 4-OHE1 by CYP1B1 (15), both of which can be subsequently methylated to 2-MeOE1 or 4-MeOE1, respectively. (17) Alternatively, CYP3A4 has high affinity in the hydroxylation of estrone to 16a-OHE1 (70), an intermediate which can ultimately produce estriol. Estriol is only present in pregnant females and represents 90% of all estrogens during pregnancy. (63) Estradiol is the main estrogen that circulates in women throughout the menstrual cycle (6) but drops off after menopause due to ovarian failure. (47) Estrone concentrations have been measured as three-fold higher than estradiol postmenopause. (6)Ingredients that affect the steroidogenic pathways
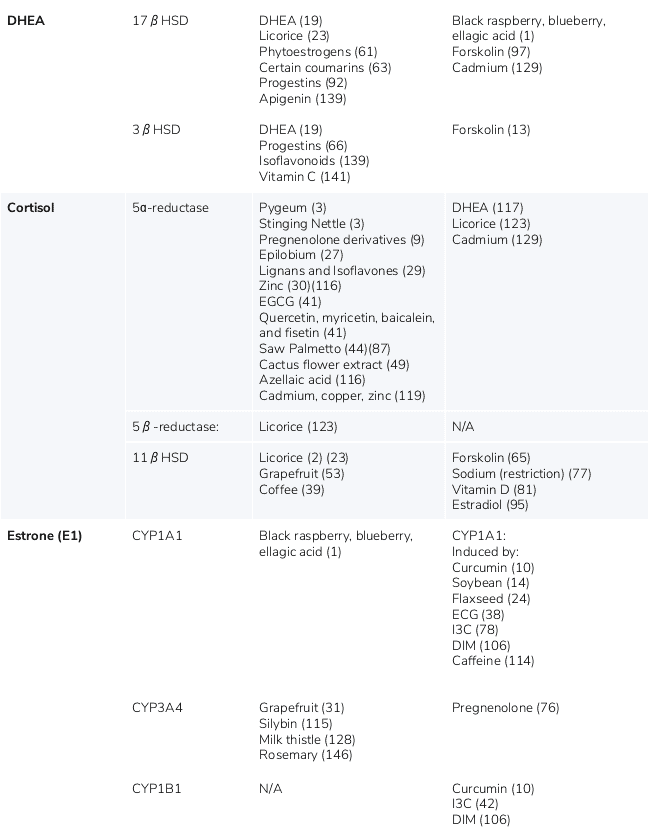
The following table provides a list of ingredients that can either increase or decrease the production of hormones produced via the steroid pathway.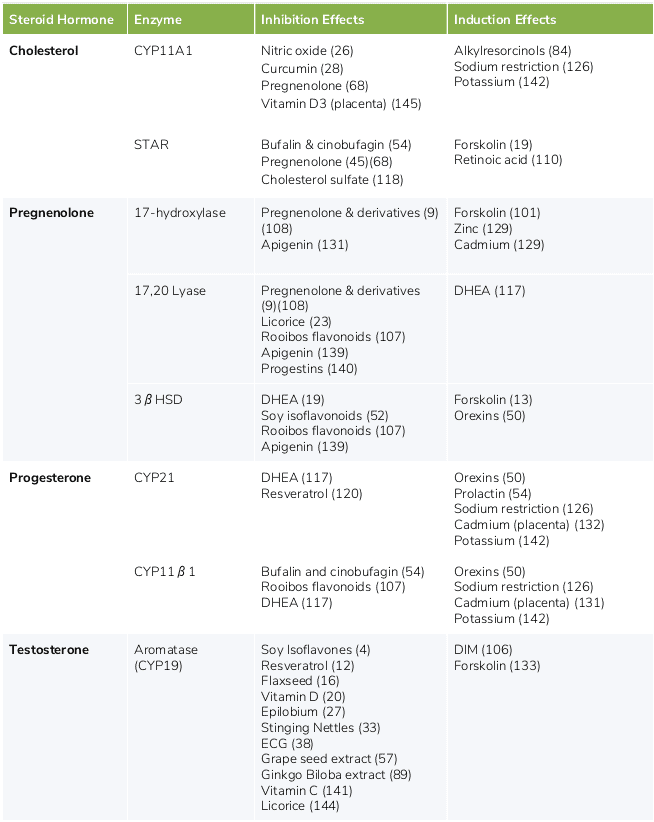
The bottom line
In summary, the steroidogenic pathways describe the production of steroid hormones from cholesterol. Several natural ingredients influence the production of steroid hormones via the steroidogenic pathways. For an in-depth look at six major natural ingredients and nutrients that affect the pathways, visit the Fullscript blog.- Adam, E. K., Quinn, M. E., Tavernier, R., Mcquillan, M. T., Dahlke, K. A., & Gilbert, K. E. (2017). Diurnal cortisol slopes and mental and physical health outcomes: A systematic review and meta-analysis. Psychoneuroendocrinology,83, 25-41.
- Aiyer, H. S., & Gupta, R. C. (2010). Berries and ellagic acid prevent estrogen-induced mammary tumorigenesis by modulating enzymes of estrogen metabolism. Cancer Prevention Research,3(6), 727-737.
- Al-Dujaili, E., Kenyon, C., Nicol, M., & Mason, J. (2011). Liquorice and glycyrrhetinic acid increase DHEA and deoxycorticosterone levels in vivo and in vitro by inhibiting adrenal SULT2A1 activity. Molecular and Cellular Endocrinology,336(1-2), 102-109.
- Allkanjari, O., & Vitalone, A. (2015). What do we know about phytotherapy of benign prostatic hyperplasia? Life Sciences,126, 42-56.
- Amaral, C., Toloi, M. R., Vasconcelos, L. D., Fonseca, M. J., Correia-Da-Silva, G., & Teixeira, N. (2017). The role of soybean extracts and isoflavones in hormone-dependent breast cancer: Aromatase activity and biological effects. Food & Function,8(9), 3064-3074.
- Baird, D. T., & Guevara, A. (1969). The menstrual cycle concentration of unconjugated estrone and estradiol in peripheral plasma in nonpregnant women throughout the menstrual cycle, castrate and postmenopausal women and in men. Obstetrical & Gynecological Survey,24(9), 1177-1179.
- Banday, A. H., Shameem, S. A., Banday, J. A., & Ganaie, B. A. (2019). Synthesis, 17α-hydroxylase-C17,20-lyase inhibitory and 5AR reductase activity of novel pregnenolone derivatives. Anti-Cancer Agents in Medicinal Chemistry,18(13), 1919-1926.
- Bandulik, S. (2016). Of channels and pumps: Different ways to boost the aldosterone? Acta Physiologica,220(3), 332-360.
- Bansal, S. S., Kausar, H., Vadhanam, M. V., Ravoori, S., Pan, J., Rai, S. N., & Gupta, R. C. (2014). Curcumin implants, not curcumin diet, inhibit estrogen-induced mammary carcinogenesis in ACI rats. Cancer Prevention Research,7(4), 456-465.
- Baravalle, R., Ciaramella, A., Baj, F., Nardo, G. D., & Gilardi, G. (2018). Identification of endocrine disrupting chemicals acting on human aromatase. Biochimica Et Biophysica Acta (BBA) – Proteins and Proteomics,1866(1), 88-96.
- Bird, I. M., Imaishi, K., Pasquarette, M. M., Rainey, W. E., & Mason, J. I. (1996). Regulation of 3 beta-hydroxysteroid dehydrogenase expression in human adrenocortical H295R cells. The Journal of Endocrinology,150, S165-S173.
- Bogacz, A., Bartkowiak-Wieczrek, J., Mikołajczak, P., Rakowska-Mrozikiewicz, B., Grześkowiak, E., Wolski, H., . . . Mrozikiewicz, P. (2014). The influence of soybean extract on the expression level of selected drug transporters, transcription factors and cytochrome P450 genes encoding phase I drug-metabolizing enzymes. Polish Gynaecology,85(5).
- Brooks, J. D., & Thompson, L. U. (2005). Mammalian lignans and genistein decrease the activities of aromatase and 17β-hydroxysteroid dehydrogenase in MCF-7 cells. The Journal of Steroid Biochemistry and Molecular Biology,94(5), 461-467.
- Chen, W., Tsai, S., Sheu, H., Tsai, J., & Zouboulis, C. C. (2010). Testosterone synthesized in cultured human SZ95 sebocytes derives mainly from dehydroepiandrosterone. Experimental Dermatology,19(5), 470-472.
- Cribb, A. E., Knight, M. J., Dryer, D., Guernsey, J., Hender, K., Tesch, M., & Saleh, T. M. (2006). Role of polymorphic human cytochrome P450 enzymes in estrone oxidation. Cancer Epidemiology Biomarkers & Prevention,15(3), 551-558.
- Cutolo, M., Paolino, S., Sulli, A., Smith, V., Pizzorni, C., & Seriolo, B. (2014). Vitamin D, steroid hormones, and autoimmunity. Annals of the New York Academy of Sciences,1317(1), 39-46.
- Dawling, S., Roodi, N., Mernaugh, R. L., Wang, X., & Parl, F. F. (2001). Catechol-O-methyltransferase (COMT)-mediated metabolism of catechol estrogens: Comparison of wild-type and variant COMT isoforms. Cancer Research,61(18), 6716-6722.
- Deluca, D., Krazeisen, A., Breitling, R., Prehn, C., Möller, G., & Adamski, J. (2005). Inhibition of 17beta-hydroxysteroid dehydrogenases by phytoestrogens: Comparison with other steroid metabolizing enzymes. The Journal of Steroid Biochemistry and Molecular Biology,93(2-5), 285-292.
- Dikshit, A., Hales, K., & Hales, D. B. (2017). Whole flaxseed diet alters estrogen metabolism to promote 2-methoxtestradiol-induced apoptosis in hen ovarian cancer. The Journal of Nutritional Biochemistry,42, 117-125.
- Drewett, J. G., Adams-Hays, R. L., Ho, B. Y., & Hegge, D. J. (2002). Nitric oxide potently inhibits the rate-limiting enzymatic step in steroidogenesis. Molecular and Cellular Endocrinology,194(1-2), 39-50.
- Ducrey, B., Marston, A., Göhring, S., Hartmann, R., & Hostettmann, K. (1997). Inhibition of 5α-reductase and aromatase by the ellagitannins oenothein A and oenothein B from Epilobium species. Planta Medica,63(2), 111-114.
- Enyeart, J. A., Liu, H., & Enyeart, J. J. (2009). Curcumin inhibits ACTH- and angiotensin II-stimulated cortisol secretion and Cav3.2 current. Journal of Natural Products,72(8), 1533-1537.
- Evans, B. A., Griffiths, K., & Morton, M. S. (1995). Inhibition of 5α-reductase in genital skin fibroblasts and prostate tissue by dietary lignans and isoflavonoids. Journal of Endocrinology,147(2), 295-302.
- Fahim, M. S., Wang, M., Sutcu, M. F., & Fahim, Z. (2009). Zinc arginine, a 5α-reductase inhibitor, reduces rat ventral prostate weight and DNA without affecting testicular function. Andrologia,25(6), 369-375.
- Fukuda, K., Ohta, T., Oshima, Y., Ohashi, N., Yoshikawa, M., & Yamazoe, Y. (1997). Specific CYP3 A4 inhibitors in grapefruit juice: Furocoumarin dimers as components of drug interaction. Pharmacogenetics,7(5), 391-396.
- Gansser, D., & Spiteller, G. (1995). Aromatase inhibitors from Urtica dioica roots. Planta Medica,61(2), 138-140.
- Garg, D., Ng, S. S., Baig, K. M., Driggers, P., & Segars, J. (2017). Progesterone-mediated non-classical signaling. Trends in Endocrinology & Metabolism,28(9), 656-668.
- Goodin, M., Bray, B., & Rosengren, R. (2006). Sex- and strain-dependent effects of epigallocatechin gallate (EGCG) and epicatechin gallate (ECG) in the mouse. Food and Chemical Toxicology,44(9), 1496-1504.
- Gumy, C., Thurnbichler, C., Aubry, E. M., Balazs, Z., Pfisterer, P., Baumgartner, L., . . . Rollinger, J. M. (2009). Inhibition of 11β-hydroxysteroid dehydrogenase type 1 by plant extracts used as traditional antidiabetic medicines. Fitoterapia,80(3), 200-205.
- Hiipakka, R. A., Zhang, H., Dai, W., Dai, Q., & Liao, S. (2002). Structure–activity relationships for inhibition of human 5α-reductases by polyphenols. Biochemical Pharmacology, 63(6), 1165-1176.
- Horn, T. L., Reichert, M. A., Bliss, R. L., & Malejka-Giganti, D. (2002). Modulations of P450 mRNA in liver and mammary gland and P450 activities and metabolism of estrogen in liver by treatment of rats with indole-3-carbinol. Biochemical Pharmacology,64(3), 393-404.
- Iehlé, C., Délos, S., Guirou, O., Tate, R., Raynaud, J., & Martin, P. (1995). Human prostatic steroid 5α-reductase isoforms—A comparative study of selective inhibitors. The Journal of Steroid Biochemistry and Molecular Biology,54(5-6), 273-279.
- Jonas, A., Rosenblat, G., Krapf, D., Bitterman, W., & Neeman, I. (1998). Cactus flower extracts may prove beneficial in benign prostatic hyperplasia due to inhibition of 5α reductase activity, aromatase activity and lipid peroxidation. Urological Research,26(4), 265-270.
- Kagerer, S. M., Eichholz, C., & Jöhren, O. (2011). Orexins/hypocretins increase the promoter activity of selective steroidogenic enzymes. Peptides,32(4), 839-843.
- Kaminska, B., Clereszko, R., Kiezun, M., & Dusza, L. (2013). In vitro effects of genistein and daidzein on the activity of adrenocortical steroidogenic enzymes in mature female pigs. Journal of Physiology and Pharmacology,64(1), 103-108.
- Kargl, C., Arshad, M., Salman, F., Schurman, R. C., & Corral, P. D. (2017). 11β-hydroxysteroid dehydrogenase type-II activity is affected by grapefruit juice and intense muscular work. Archives of Endocrinology and Metabolism,61(6), 556-561.
- Kau, M., Lo, M., Tsai, S., Chen, J., Pu, H., Chien, E. J., . . . Wang, P. S. (1999). Effects of prolactin on aldosterone secretion in rat zona glomerulosa cells. Journal of Cellular Biochemistry,72(2), 286-293.
- Kau, M., Wang, J., Tsai, S., Yu, C., & Wang, P. S. (2012). Inhibitory effect of bufalin and cinobufagin on steroidogenesis via the activation of ERK in human adrenocortical cells. British Journal of Pharmacology,165(6), 1868-1876.
- Kennedy, R. G., Davies, T., & Al-Azzawi, F. (1997). Sexual interest in postmenopausal women is related to 5alpha-reductase activity. Human Reproduction,12(2), 209-213.
- Kijima, I., Phung, S., Hur, G., Kwok, S., & Chen, S. (2006). Grape seed extract is an aromatase inhibitor and a suppressor of aromatase expression. Cancer Research,66(11), 5960-5967.
- Krazeisen, A., Breitling, R., Möller, G., & Adamski, J. (2002). Human 17β-hydroxysteroid dehydrogenase type 5 is inhibited by dietary flavonoids. Flavonoids in Cell Function Advances in Experimental Medicine and Biology,505, 151-161.
- Lain, R. L., Barrell, K., Saeed, G., Nicholls, P., Simons, C., Kirby, A., & Smith, H. (2002). Some coumarins and triphenylethene derivatives as inhibitors of human testes microsomal 17β-hydroxysteroid dehydrogenase (17β-HSD type 3): Further studies with tamoxifen on the rat testes microsomal enzyme. Journal of Enzyme Inhibition and Medicinal Chemistry,17(2), 93-100.
- Lecybyl, R., Rubis, B., Krozowski, Z., & Trzeciak, W. H. (2003). Regulation of 11β‐hydroxysteroid dehydrogenase type II expression in the renal epithelial cells. Endocrine Research,29(2), 211-216.
- Lee, T. C., Miller, W. L., & Auchus, R. J. (1999). Medroxyprogesterone acetate and dexamethasone are competitive inhibitors of different human steroidogenic enzymes. The Journal of Clinical Endocrinology & Metabolism, 84(6), 2104-2110.
- Li, J., Zhou, Q., Ma, Z., Wang, M., Shen, W., Azhar, S., . . . Hu, Z. (2017). Feedback inhibition of CREB signaling by p38 MAPK contributes to the negative regulation of steroidogenesis. Reproductive Biology and Endocrinology,15(1).
- Manna, P. R., Stetson, C. L., Slominski, A. T., & Pruitt, K. (2015). Role of the steroidogenic acute regulatory protein in health and disease. Endocrine,51(1), 7-21.
- Mansur, A. D., Silva, T. C., Takada, J. Y., Avakian, S. D., Strunz, C. M., César, L. A., . . . Ramires, J. A. (2012). Long-term prospective study of the influence of estrone levels on events in postmenopausal women with or at high risk for coronary artery disease. The Scientific World Journal,2012, 1-6.
- Marks, L. S. (2004). 5α-reductase: History and clinical importance. Reviews in Urology,6(9), S11-S21.
- Masuyama, H., Nakamura, K., Nobumoto, E., & Hiramatsu, Y. (2016). Inhibition of pregnane X receptor pathway contributes to the cell growth inhibition and apoptosis of anticancer agents in ovarian cancer cells. International Journal of Oncology,49(3), 1211-1220.
- McBride, J. A., Carson, C. C., & Coward, R. M. (2015). Diagnosis and management of testosterone deficiency. Asian Journal of Andrology,17(2), 177-186.
- Mckinnell, J., Roscoe, D., Holmes, M. C., Lloyd-Macgilp, S. A., & Kenyon, C. J. (2000). Regulation of 11β-hydroxysteroid dehydrogenase enzymes by dietary sodium in the rat. Endocrine Research,26(1), 81-95.
- Michnovicz, J. J., & Bradlow, H. L. (1990). Induction of estradiol metabolism by dietary indole-3-carbinol in humans. JNCI Journal of the National Cancer Institute,82(11), 947-949.
- Miller, W. L., & Auchus, R. J. (2011). The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocrine Reviews,32(1), 81-151.
- Miller, W. L. (2017). Steroidogenesis: Unanswered questions. Trends in Endocrinology & Metabolism,28(11), 771-793.
- Morris, K. L., & Zemel, M. B. (2005). 1, 25-dihydroxyvitamin D3 modulation of adipocyte glucocorticoid function. Obesity Research,13(4), 670-677.
- Oskarsson, A., & Andersson, Å O. (2016). Suppressed sex hormone biosynthesis by alkylresorcinols: A possible link to chemoprevention. Nutrition and Cancer,68(6), 978-987.
- Pais, P. (2010). Potency of a novel saw palmetto ethanol extract, SPET-085, for inhibition of 5α-reductase II. Advances in Therapy,27(8), 555-563.
- Park, Y. J., Choo, W. H., Kim, H. R., Chung, K. H., & Oh, S. M. (2015). Inhibitory aromatase effects of flavonoids from ginkgo biloba extracts on estrogen biosynthesis. Asian Pacific Journal of Cancer Prevention,16(15), 6317-6325.
- Pasqualini, J. R., Ebert, C., & Chetrite, G. S. (2001). Biological effects of progestins in breast cancer. Gynecological Endocrinology,15(6), 44-52.
- Pepe, G. J., Davies, W. A., Dong, K., Luo, H., & Albrecht, E. D. (1999). Cloning of the 11β-hydroxysteroid dehydrogenase (11β-HSD)-2 gene in the baboon: Effects of estradiol on promoter activity of 11β-HSD-1 and -2 in placental JEG-3 cells. Biochimica Et Biophysica Acta (BBA) – Gene Structure and Expression,1444(1), 101-110.
- Prough, R. A., Clark, B. J., & Klinge, C. M. (2016). Novel mechanisms for DHEA action. Journal of Molecular Endocrinology,56(3), R139-R155.
- Qin, K., & Rosenfield, R. L. (2005). Characterization of the basal promoter element of the human type 5 17beta-hydroxysteroid dehydrogenase gene. Biochimica Et Biophysica Acta (BBA) – Gene Structure and Expression,1728(3), 115-125.
- Robinson, D. P., & Klein, S. L. (2012). Pregnancy and pregnancy-associated hormones alter immune responses and disease pathogenesis. Hormones and Behavior,62(3), 263-271.
- Roelofs, M. J., Piersma, A. H., Berg, M. V., & Duursen, M. B. (2013). The relevance of chemical interactions with CYP17 enzyme activity: Assessment using a novel in vitro assay. Toxicology and Applied Pharmacology,268(3), 309-317.
- Sanderson, J. T., Slobbe, L., Lansbergen, G. W., Safe, S., & Van den Berg, M. (2001). 2,3,7,8-Tetrachlorodibenzo-p-dioxin and diindolylmethanes differentially induce cytochrome P450 1A1, 1B1, and 19 in H295R human adrenocortical carcinoma cells. Toxicological Sciences,61(1), 40-48.
- Sasano, H., & Sasano, N. (1988). Extraadrenal immunohistochemical distribution of steroid 21-hydroxylase in human. The Tohoku Journal of Experimental Medicine,154(1), 21-28.
- Schloms, L., & Swart, A. (2014). Rooibos flavonoids inhibit the activity of key adrenal steroidogenic enzymes, modulating steroid hormone levels in H295R cells. Molecules,19(3), 3681-3695.
- Schoonen, W. G., Lambert, J. G., Resink, J. W., Viveen, W. J., & Van Oordt, P. G. (1987). A quantitative study of steroid bioconversions in the testis of the African catfish, Clarias gariepinus (Burchell), under natural spawning and natural and cultivated non-spawning conditions. Journal of Endocrinology,112(2), 323-332.
- Sesta, A., Cassarino, M. F., Tapella, L., Castelli, L., Cavagnini, F., & Giraldi, F. P. (2016). Effect of retinoic acid on human adrenal corticosteroid synthesis. Life Sciences,151, 277-280.
- Shou, M., Korzekwa, K. R., Brooks, E. N., Krausz, K. W., Gonzalez, F. J., & Gelboin, H. V. (1997). Role of human hepatic cytochrome P450 1A2 and 3A4 in the metabolic activation of estrone. Carcinogenesis,18(1), 207-214.
- Sowers, M. R., Crawford, S., Mcconnell, D. S., Randolph, J. F., Gold, E. B., Wilkin, M. K., & Lasley, B. (2006). Selected diet and lifestyle factors are associated with estrogen metabolites in a multiracial/ethnic population of women. The Journal of Nutrition,136(6), 1588-1595.
- Sridar, C., Goosen, T. C., Kent, U. M., Williams, J. A., & Hollenberg, P. F. (2004). Silybin inactivates cytochromes P450 3A4 and 2C9 and inhibits major hepatic glucuronosyltransferases. Drug Metabolism and Disposition,32(6), 587-594.
- Stamatiadis, D., Bulteau-Portois, M., & Mowszowicz, I. (1988). Inhibition of 5α-reductase activity in human skin by zinc and azelaic acid. British Journal of Dermatology,119(5), 627-632.
- Stomati, M., Monteleone, P., Casarosa, E., Quirici, B., Puccetti, S., Bernardi, F., . . . Genazzani, A. R. (2000). Six-month oral dehydroepiandrosterone supplementation in early and late postmenopause. Gynecological Endocrinology,14(5), 342-363.
- Sugawara, T., Nomura, E., & Hoshi, N. (2007). Cholesterol sulphate affects production of steroid hormones by reducing steroidogenic acute regulatory protein level in adrenocortical cells. Journal of Endocrinology,195(3), 451-458.
- Sugimoto, Y., López-Solache, I., Labrie, F., & Luu-The, V. (1995). Cations inhibit specifically type I 5α-reductase found in human skin. Journal of Investigative Dermatology,104(5), 775-778.
- Supornsilchai, V., Svechnikov, K., Seidlova-Wuttke, D., Wuttke, W., & Söder, O. (2005). Phytoestrogen resveratrol suppresses steroidogenesis by rat adrenocortical cells by inhibiting cytochrome P450 c21-hydroxylase. Hormone Research in Paediatrics,64(6), 280-286.
- Swerdloff, R. S., Dudley, R. E., Page, S. T., Wang, C., & Salameh, W. A. (2017). Dihydrotestosterone: Biochemistry, physiology, and clinical implications of elevated blood levels. Endocrine Reviews,38(3), 220-254.
- Tamura, Y., Nishikawa, T., Yamada, K., Yamamoto, M., & Kumagai, A. (1979). Effects of glycyrrhetinic acid and its derivatives on delta 4-5 alpha- and 5 beta-reductase in rat liver. Arzneimittel-Forschung,29(4), 647-649.
- Tremblay, A., Waterman, M. R., Parker, K. L., & Lehoux, J. G. (1991). Regulation of rat adrenal messenger RNA and protein levels for cytochrome P-450s and adrenodoxin by dietary sodium depletion or potassium intake. The Journal of Biological Chemistry,266(4), 2245-2251.
- Venkataramanan, R., Ramachandran, V., Komoroski, B. J., Zhang, S., Schiff, P. L., & Strom, S. C. (2000). Milk thistle, a herbal supplement, decreases the activity of CYP3A4 and uridine diphosphoglucuronosyl transferase in human hepatocyte cultures. Drug Metabolism and Disposition,28(11), 1270-1273.
- Voogt, P., Besten, P. D., Kusters, G., & Messing, M. (1987). Effects of cadmium and zinc on steroid metabolism and steroid level in the sea star Asterias rubens L. Comparative Biochemistry and Physiology Part C: Comparative Pharmacology,86(1), 83-89.
- Wang, F., Zhang, Q., Zhang, X., Luo, S., Ye, D., Guo, Y., . . . Huang, Y. (2014). Preeclampsia induced by cadmium in rats is related to abnormal local glucocorticoid synthesis in placenta. Reproductive Biology and Endocrinology,12(1), 77.
- Wang, X., Wang, G., Li, X., Liu, J., Hong, T., Zhu, Q., . . . Ge, R. (2016). Suppression of rat and human androgen biosynthetic enzymes by apigenin: Possible use for the treatment of prostate cancer. Fitoterapia,111, 66-72.
- Watanabe, M., & Nakajin, S. (2004). Forskolin up-regulates aromatase (CYP19) activity and gene transcripts in the human adrenocortical carcinoma cell line H295R. Journal of Endocrinology,180(1), 125-133.
- Wong, C., & Keung, W. (1999). Bovine adrenal 3β-hydroxysteroid dehydrogenase (E.C. 1.1.1.145)/5-ene-4-ene isomerase (E.C. 5.3.3.1): Characterization and its inhibition by isoflavones. The Journal of Steroid Biochemistry and Molecular Biology,71(5-6), 191-202.
- Woolveridge, I., & Peddie, M. J. (1997). The inhibition of androstenedione production in mature thecal cells from the ovary of the domestic hen (Gallus domesticus): Evidence for the involvement of progestins. Steroids,62(2), 214-220.
- Wu, X., Iguchi, T., Itoh, N., Okamoto, K., Takagi, T., Tanaka, K., & Nakanishi, T. (2008). Ascorbic acid transported by sodium-dependent vitamin C transporter 2 stimulates steroidogenesis in human choriocarcinoma cells. Endocrinology,149(1), 73-83.
- Yagci, A., Oertle, M., Seiler, H., Schmid, D., Campofranco, C., & Muller, J. (1996). Potassium induces multiple steroidogenic enzymes in cultured rat zona glomerulosa cells. Endocrinology,137(6), 2406-2414.
- Ye, L., Gho, W. M., Chan, F. L., Chen, S., & Leung, L. K. (2009). Dietary administration of the licorice flavonoid isoliquiritigenin deters the growth of MCF-7 cells overexpressing aromatase. International Journal of Cancer,124(5), 1028-1036.
- Zabul, P., Wozniak, M., Slominski, A., Preis, K., Gorska, M., Korozan, M., . . . Knap, N. (2015). A proposed molecular mechanism of high-dose vitamin D3 supplementation in prevention and treatment of preeclampsia. International Journal of Molecular Sciences,16(12), 13043-13064.
- Zhu, B., Loder, D. P., Cai, M. X., Ho, C. T., Huang, M. T., & Conney, A. H. (1998). Dietary administration of an extract from rosemary leaves enhances the liver microsomal metabolism of endogenous estrogens and decreases their uterotropic action in CD-1 mice. Carcinogenesis,19(10), 1821-1827.





