A resting adult’s respiration rate is between 12 and 20 respirations per minute. (5) The lungs are methodical in their rhythmic pursuit of oxygen and release of carbon dioxide. Their primary task is to bring in fresh air to oxygenate the cells of the body while removing harmful gases. The lungs also help protect us from toxins by coughing up substances so they can be properly eliminated from the body. (1)(5) Different nutrients and lifestyle factors can have a positive effect on lung health for those with and without lung disease. Keep reading to learn more.

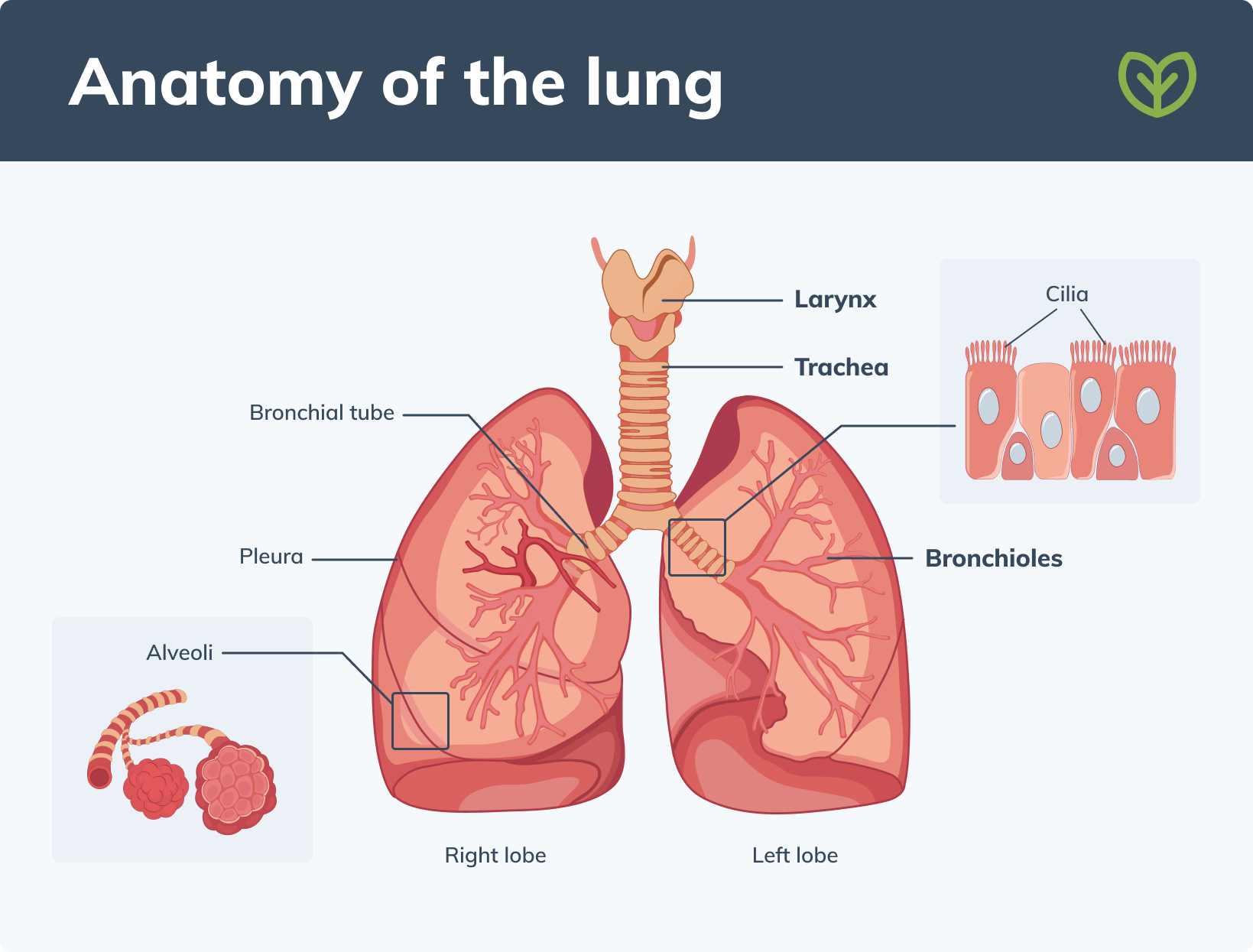
How are the lungs structured?
Physically, the lungs are divided into lobes that are surrounded by a membrane known as the pleura. The bronchial tubes within the lungs are lined with cilia that move the mucus out of the lungs. The mucus is important because it gathers up dust, germs, and any other undesirable material. At the end of the bronchioles tubes, there are small sacs called alveoli where oxygen and carbon dioxide are exchanged. (1)
The lungs are also a location in the body where a large amount of human disease can occur. (7)
Respiratory conditions
Respiratory illnesses can significantly impact quality of life and can lead to disability and even death. According to the Forum of International Respiratory Societies, the five most common and severe respiratory conditions worldwide are:
- Chronic obstructive pulmonary disease (COPD)
- Respiratory tract infections
- Tuberculosis (TB)
- Lung cancer
- Asthma (11)
Other non-infectious lung diseases include:
- Pulmonary fibrosis: This condition is a type of interstitial lung disease and is characterized by scarring of the lung tissue.
- Pulmonary hypertension: This condition is associated with elevated blood pressure in the pulmonary circulation. There are different types of pulmonary hypertension, including chronic thromboembolic pulmonary hypertension, pulmonary hypertension from left-sided heart and lung diseases, and pulmonary arterial hypertension.
- Sarcoidosis: Sarcoidosis is characterized by chronic inflammation that can impact many organs including the lungs. The chronic inflammation can result in the formation of nodules (granulomas) in the afflicted organs.
- Pleurisy: Pleurisy is characterized by inflammation of the thin layers of tissue that cover the lungs called pleura.
- Cystic fibrosis: Cystic fibrosis is a genetic disorder characterized by the production of thick mucus that can block airways and cause breathing difficulties and lung damage.
- Sleep-disordered breathing (apnea or hypopnea): Sleep apnea occurs during sleep and when upper airways in the lungs collapse due to throat muscles relaxing, resulting in blocked airflow. (10)
Did you know? COPD includes emphysema and chronic bronchitis and is a non-infectious disease characterized by lung damage that makes it hard for a person to breathe. It is one of the top leading causes of death worldwide. (11)
Respiratory support
Through a combination of diet, lifestyle, and dietary supplements, we can protect and enhance lung health and reduce the risk of non-infectious lung conditions. (16)
Diet
Air pollution, infection, and tobacco smoke contribute to oxidative stress and inflammation that can lead to COPD. When it comes to lung health, a lot can be accomplished with diet. Studies show that people with COPD often eat an inflammatory diet that does not include enough fruits and vegetables. (13) Higher intake of fruits and vegetables, in general, may help reduce the risk of certain respiratory conditions such as COPD or asthma due to the role of antioxidants in reducing oxidative stress and associated inflammation. (3)(17)
With an emphasis on fruits and vegetables as well as healthy fats, it’s not surprising that the Mediterranean diet has been shown to enhance lung health even among smokers. (5) When the Mediterranean diet is combined with exercise, it has more benefits and has been shown to improve overall lung function in healthy adults. (6)

Lifestyle
Lifestyle factors that may improve lung function include ensuring good indoor air quality in your home or building, and getting regular exercise. (2)(8)
Of course, it is well known that the single most dangerous lifestyle factor to lung health is smoking, and it is the leading cause of COPD and lung cancer. Not smoking, or stopping, is paramount when it comes to reducing the risk of non-infectious lung diseases. (16)
To improve indoor air quality, ensure that your home or building has proper ventilation, consider investing in an air purification system, keep the building clean by regularly vacuuming and mopping up contaminates, and repair any leaks or areas where moisture could accumulate. (8) Exposure to both indoor and outdoor air pollution can negatively affect lung health and checking your city’s local air quality index can be helpful for preventing exposure to outdoor air pollution.
Regular exercise can have a positive effect on lung health. Greater cardiorespiratory fitness at a young age and into adulthood may be associated with less decline of lung function over time. (2) Breathing exercises, which are a more passive form of exercise specific to the mind-body connection, may also improve pulmonary function. A meta-analysis that examined the effects of breathing exercises on individuals with lung cancer following surgery determined that breathing exercises may significantly improve quality of life and post-operative pulmonary function for individuals with the disease. (9)
Dietary supplements
Although there’s evidence that certain vitamins can be helpful for improving symptoms of COPD, it is unclear which form is most effective, supplemental or dietary. According to a 2010 systematic review, supplemental vitamins C, D, E, and A were all shown to help improve symptoms of COPD thanks to their anti-inflammatory action. (15) A 2018 review showed that vitamin A deficiency is a contributing factor for many lung diseases including COPD, lung fibrosis, lung cancer, and asthma. (14)
Other nutrients such as omega-3 fatty acids, anthocyanins, curcumin, and catechins may also support lung health and reduce the risk of lung diseases such as COPD thanks to their antioxidant and anti-inflammatory effects. (18)
Many medicinal herbs possess anti-inflammatory properties which help support,protect, and enhance lung function. In addition to curcumin found in turmeric and catechins found in green tea, other herbs and plant components that have been shown to reduce the risk and the symptoms of COPD and asthma include luteolin, quercetin, apigenin, and astragalus. (12)
The bottom line
Lung diseases can dramatically and negatively impact the quality of life. They can also be life-threatening. That’s why taking proactive steps to prevent these respiratory illnesses is worth the effort. We can enhance our lung health through a combination of diet, lifestyle, and supplementation when necessary. Speak to your integrative healthcare practitioner before making changes to your health routine.
- Alvarado, A., & Arce, I. (2016). Metabolic functions of the lung, disorders and associated pathologies. Journal of Clinical Medicine Research, 8(10), 689–700.
- Benck, L. R., Cuttica, M. J., Colangelo, L. A., Sidney, S., Dransfield, M. T., Mannino, D. M., Jacobs, D. R., Lewis, C. E., Zhu, N., Washko, G. R., Liu, K., Carnethon, M. R., & Kalhan, R. (2017). Association between cardiorespiratory fitness and lung health from young adulthood to middle age. American Journal of Respiratory and Critical Care Medicine, 195(9), 1236–1243.
- Berthon, B., & Wood, L. (2015). Nutrition and respiratory Health—Feature review. Nutrients, 7(3), 1618–1643.
- Chaudhry, R., & Bordoni, B. (2021). Anatomy, thorax, lungs . StatPearls Publishing.
- Chourpiliadis, C., & Bhardwaj, A. (2021). Physiology, respiratory rate . StatPearls Publishing.
- Gutiérrez-Carrasquilla, L., Sánchez, E., Hernández, M., Polanco, D., Salas-Salvadó, J., Betriu, N., Gaeta, A., Carmona, P., Purroy, F., Pamplona, R., Farràs, C., López-Cano, C., Fernández, E., & Lecube, A. (2019). Effects of mediterranean diet and physical activity on pulmonary function: A Cross-Sectional analysis in the ILERVAS project. Nutrients, 11(2), 329.
- Haddad, M., & Sharma, S. (2021). Physiology, lung . StatPearls Publishing.
- Health Canada. (2018). Infographic: Maintain and improve indoor air quality. Government of Canada. https://www.canada.ca/en/health-canada/services/publications/healthy-living/infographic-improve-indoor-air-quality.html
- LIU, W., PAN, Y. L., GAO, C. X., SHANG, Z., NING, L. J., & LIU, X. (2013). Breathing exercises improve post-operative pulmonary function and quality of life in patients with lung cancer: A meta-analysis. Experimental and Therapeutic Medicine, 5(4), 1194–1200.
- Lung disease lookup. (n.d.). American Lung Association. https://www.lung.org/lung-health-diseases/lung-disease-lookup
- Marciniuk, D. D., Schraufnagel, D. E., & European Respiratory Society. (2017). The global impact of respiratory disease. European Respiratory Society.
- Santana, F. P. R., Pinheiro, N. M., Mernak, M. I. B., Righetti, R. F., Martins, M. A., Lago, J. H. G., Lopes, F. D. T. Q. D. S., Tibério, I. F. L. C., & Prado, C. M. (2016). Evidences of herbal Medicine-Derived natural products effects in inflammatory lung diseases. Mediators of Inflammation, 2016, 1–14.
- Scoditti, E., Massaro, M., Garbarino, S., & Toraldo, D. M. (2019). Role of diet in chronic obstructive pulmonary disease prevention and treatment. Nutrients, 11(6), 1357.
- Timoneda, J., Rodríguez-Fernández, L., Zaragozá, R., Marín, M., Cabezuelo, M., Torres, L., Viña, J., & Barber, T. (2018). Vitamin a deficiency and the lung. Nutrients, 10(9), 1132.
- Tsiligianni, I. G., & van der Molen, T. (2010). A systematic review of the role of vitamin insufficiencies and supplementation in COPD. Respiratory Research, 11(1).
- West, R. (2017). Tobacco smoking: Health impact, prevalence, correlates and interventions. Psychology & Health, 32(8), 1018–1036.
- Wood, L. G., Garg, M. L., Smart, J. M., Scott, H. A., Barker, D., & Gibson, P. G. (2012). Manipulating antioxidant intake in asthma: A randomized controlled trial. The American Journal of Clinical Nutrition, 96(3), 534–543.
- Zhai, T., Li, S., Hu, W., Li, D., & Leng, S. (2018). Potential micronutrients and phytochemicals against the pathogenesis of chronic obstructive pulmonary disease and lung cancer. Nutrients, 10(7), 813.





